|
Back to Blog
Van der waals equation calculator5/15/2023  
Theertharaman, " A New Four-Parameter Generalized van der Waals Equation of State: Metastable State of Group IV Elements", International Journal of Science and Research (IJSR), Volume 7 Issue 4, April 2018, pp. 
Type Your Email Address below to Download the Article PDF Keywords: Equation of state, Law of corresponding states, Germanium, Lead, Silicon, Tin, Spinodal, Superheating, Use the van der Waals equation and the ideal gas equation to calculate the volume of 1.000 mol of neon at a pressure of 500.0 atm and a temperature of 355.0 K. This fact is to be taken into account when silicon, germanium, tin and lead are subjected to rapid heating. The van der Waals equation is an equation of state which extends the ideal gas law and includes the intermolecular forces of attraction between the molecules. Calculate the pressures given by the ideal gas equation and the van der Waals equation. Above these temperatures, silicon, germanium, tin and lead undergo explosive boiling by virtue of homogeneous nucleation. Example 1.3 At T300 K, 1.00 mol of CO2 occupies a volume of 1.50 L. It has been established that silicon, germanium, tin and lead can be superheated, under rapid heating, up to temperatures 4481K, 7975K, 7161K, and 4333K, respectively. It is established that the new parameter introduced in the attractive term of the equation of state is a thermodynamic similarity parameter. It is established that silicon, germanium, tin and lead obey the single parameter law of corresponding states. Use the van der Waals equation and the ideal gas equation to calculate the volume of 1.000 mol of neon at a pressure of 500.0 atm and a temperature of 355.0 K. The physical process that leads to Van der Waals interactions is clear, but it is difficult to determine the precise parameters that would allow us to calculate the energies of Van der Waals. How would you use the van der Waals equation of state to calculate the pressure of 3.60 mol of H2O at 453 K in a 5. Even macroscopic surfaces experience VDW interactions, but more of this later. 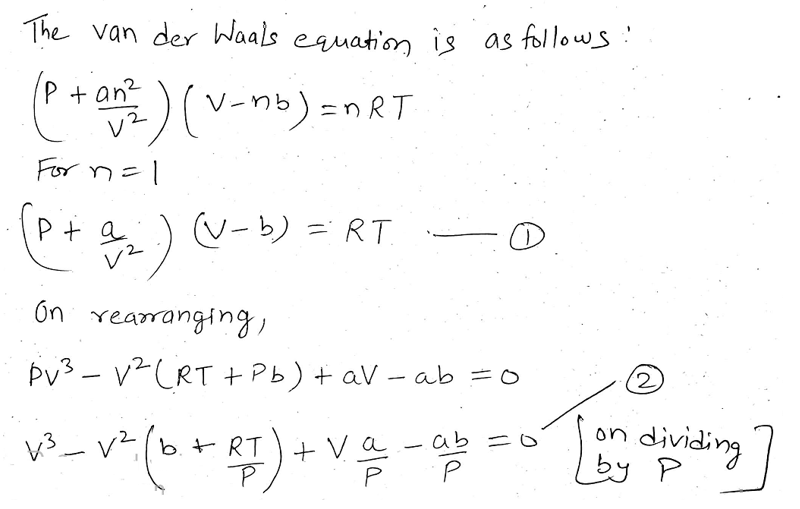
Research Paper | Physics and Astronomy | India | Volume 7 Issue 4, April 2018Ī New Four-Parameter Generalized van der Waals Equation of State: Metastable State of Group IV ElementsĪbstract: A new four- parameter generalized van der Waals equation of state has been proposed and employed to calculate the spinodal (Metastable State) and the thermodynamic limit of superheat of the of silicon, germanium, lead and tin. Any two molecules experience Van der Waals interactions. The reason for dividing by 2 rather than some other constant is still somewhat unclear, but the overlap explanation at least shows why multiplying $N$ by the volume of a sphere of radius $\sigma$ would be overcounting.Google Scholar CrossRef Member CiteFactor Index ScienceGate Index Scite_ Index Digital Repository Index Noel Meorial Library Index Academia.Edu Index If we consider a cube of volume V, then we can say that the side of this cube is of length $V^\pi\sigma^3$$ When accounting for the excluded volume for in the Van der Waals equation, it is assumed that the molecules are hard spheres and are of diameter. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
